FGFR alterations and tumour pathogenesis

The FGFR signalling pathway
Fibroblast growth factor receptors (FGFRs) belong to a family of four transmembrane receptor tyrosine kinases (FGFR1–4) that help regulate multiple physiological processes.[1] They are highly conserved, widely distributed receptors and activate numerous physiological processes that can also be associated with tumour growth.[1][2]
In the fibroblast growth factor (FGF)/FGFR signalling pathway (Figure 1), FGF ligand binding triggers autophosphorylation of FGFR.[3] Docking proteins such as FGFR substrate 2 (FRS2a) and PLCγ then activate downstream pathways, including RAS/RAF/MEK, PI3K/AKT/mTOR, and STAT.[3]
Figure 1. The FGF/FGFR signalling pathway
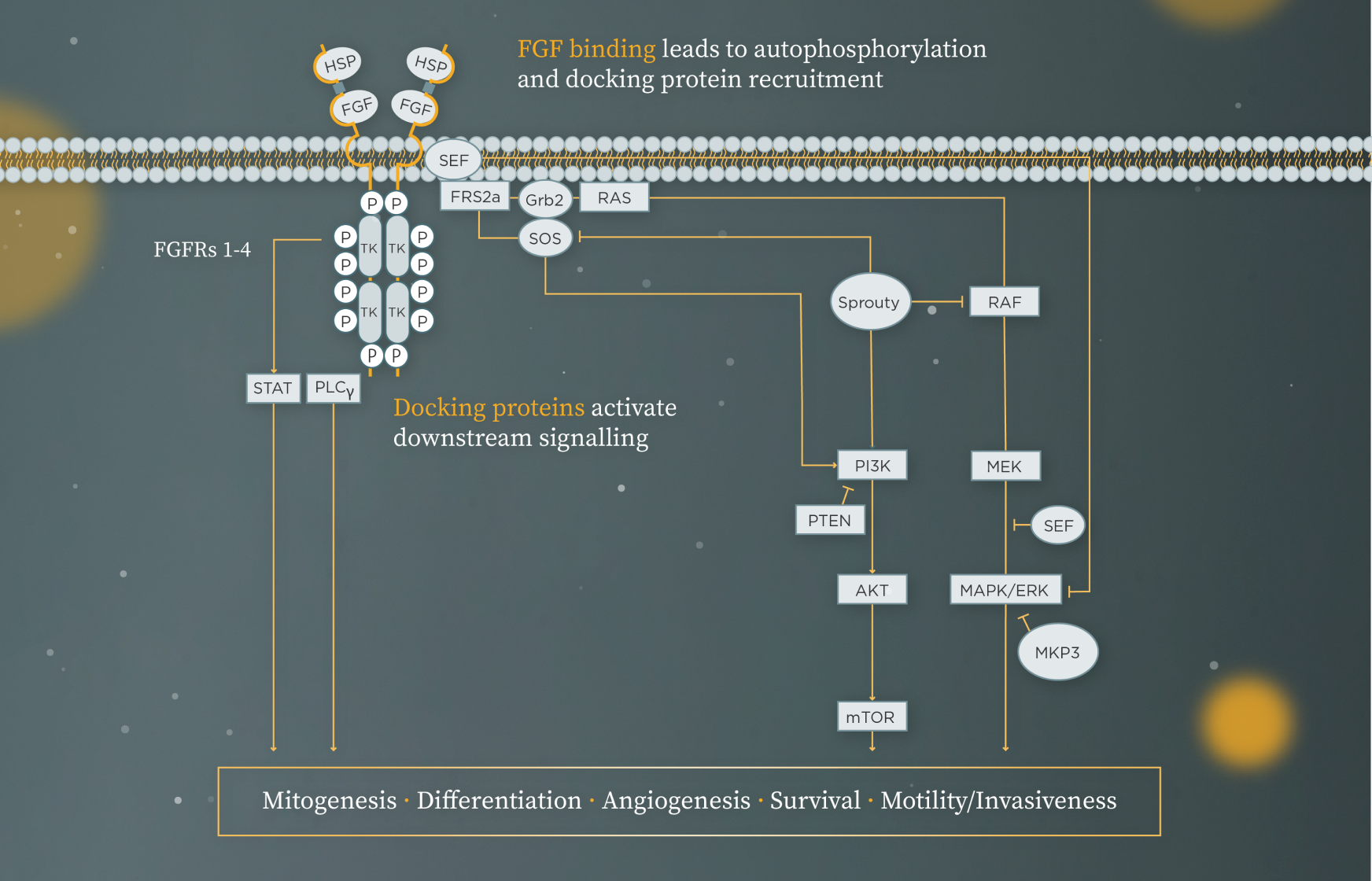
Adapted from Corn PG et al. 2013 and Yang et al. 2019.[3][4]
Figure legend can be found in footnotes at the bottom of the page.
Types of FGFR alterations and mechanisms of tumourigenesis
The majority of FGFR genetic alterations lead to gain-of-function and include:[1][2]
- Activating FGFR mutations in the extracellular, transmembrane or tyrosine kinase domain of the receptor
- Chromosomal rearrangements
- FGFR gene amplification, translocation or transcriptional deregulation
Aberrant FGFR signalling contributes to oncogenesis through several ligand-dependent and -independent mechanisms (Figure 2):[5]
- Protein overexpression due to FGFR gene amplification
- Ligand-independent dimerisation of FGFRs due to activating mutations
- Ligand-independent receptor hyperactivation due to chromosomal translocations
- Autocrine or paracrine signalling due to FGFs produced by tumour cells and the stromal compartment, respectively
- Angiogenesis or epithelial–mesenchymal transition (EMT) due to tumour cell FGF secretion
- Hyperactivation of the FGFR downstream signalling pathways due to gene amplification or protein overexpression of FGFR binding partners
Figure 2. Mechanisms underlying FGFR tumourigenesis
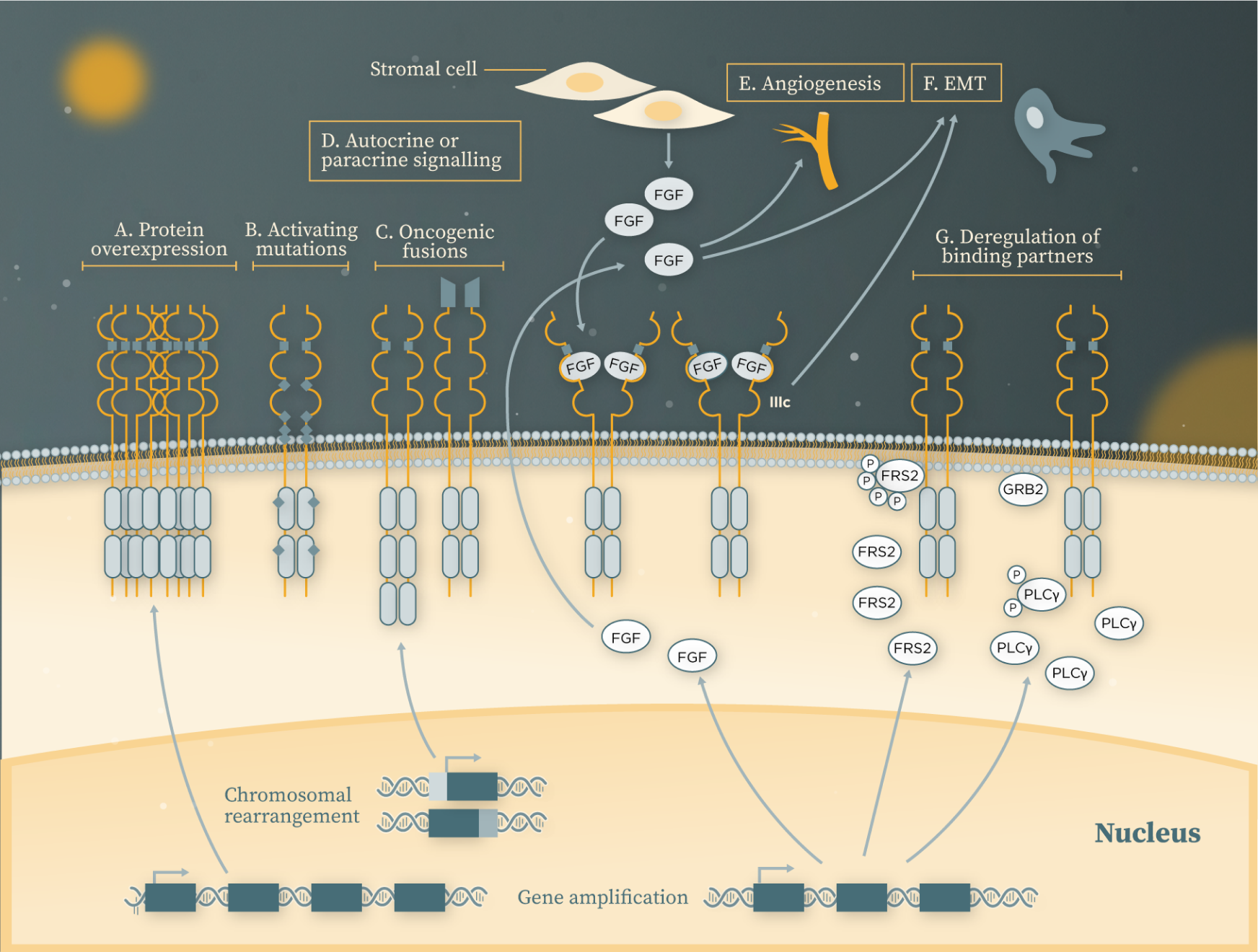
Adapted from Babina IS and Turner NC. 2017.[5]
Figure legend can be found in footnotes at the bottom of the page.
Different cancers associated with FGFR alterations
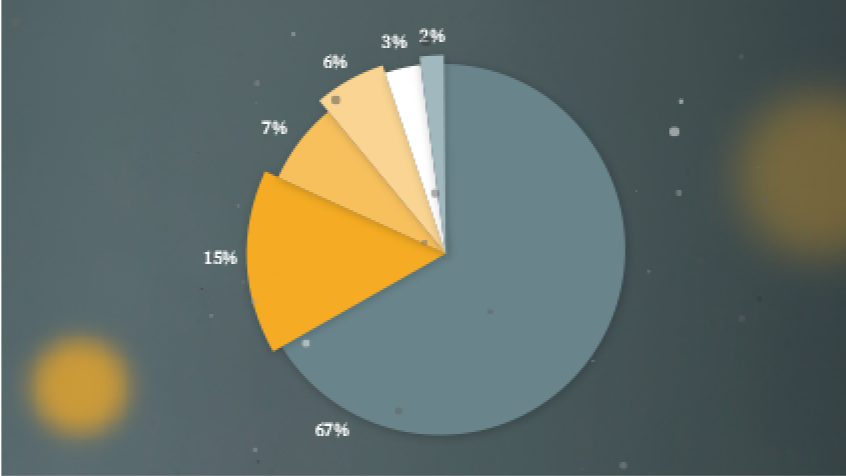
FGFR alterations are common in a wide variety of cancers and represent an important potential target across tumour types.[1] In an analysis of 4,853 cancers, those that commonly harbored FGFR alterations included:*,[6]
- 31.7% of urothelial
- 17.4% of breast
- 11.3% of endometrial
- 8.6% of ovarian
- 7.6% of glioma
- 7.0% of cholangiocarcinoma
- 6.7% of gastric/gastroesophageal junction
- 5.2% of non-small cell lung cancer
- 4.7% of pancreatic exocrine
- 4.6% of cancer of the head and neck
- 4.0% of sarcoma
The type of FGFR alterations most commonly found can differ in different cancer types:[1]
- FGFR1 amplifications predominate in squamous cell lung, breast, ovarian, and urothelial cancers
- FGFR3 mutations predominate in bladder and other urothelial tumours
The majority of FGFR alterations observed in tumours to date lead to gain-of-function[1]

Explore more
Learn more about FGFR alterations in oncology and their importance in UC:

Footnotes
Figure 1 Legend
AKT=protein kinase B; ERK=extracellular signal-regulated kinase; FGF=fibroblast growth factor; FRS2a=FGF receptor substrate 2a; GRb2=growth factor receptor bound protein 2; HSP=heparan sulfate proteoglycans; MAPK=mitogen-activated protein kinase; MEK=MAPK/ERK kinase; MKP3=mitogen-activated protein kinase phosphatase 3; mTOR=mammalian target of rapamycin; P=phosphate; PI3K=phosphoinositide 3-kinase; PLCγ=phospholipase C gamma; RAF=rapidly accelerated fibrosarcoma kinases; RAS=retrovirus-associated DNA sequences; SEF=spatial-temporal regulator of MAPK signalling; SOS=Son of Sevenless; STAT=signal transducer and activator of transcription; TK=tyrosine kinase domain.
Figure 2 Legend
A. FGFR gene amplification often translates into protein overexpression, leading to increased receptor accumulation and activation of the downstream signalling pathways.
B. Activating mutations often result in increased dimerisation of the receptors in the absence of ligand, or constitutive activation of the kinase domain.
C. As a result of chromosomal translocations, parts of FGFRs may become fused with genes encoding other proteins at either carboxy or amino termini, thereby either increasing dimerisation of the receptors or falling under the promoter regions of a different protein, resulting in receptor hyperactivation in a ligand-independent manner.
D. FGFRs can be overstimulated by their ligands in an autocrine fashion, where fibroblast growth factors (FGFs) are produced by the tumour cells, or by paracrine signalling, where FGFs are secreted by the stromal compartment. In response to a stimulus, or as a result of gene amplification, the third immunoglobulin (Ig) III loop can also be alternatively spliced from the IIIb to the IIIc isoform, which alters the ligand specificity and affinity of the receptors, resulting in altered autocrine signalling.
E. FGFs secreted by the tumour cells, or tumour-associated stromal cells may contribute to angiogenesis.
F. FGFs secreted by the tumour cells, or tumour-associated stromal cells may contribute to epithelial–mesenchymal transition (EMT).
G. Deregulation of the FGFR binding partners FGFR substrate 2 (FRS2) and phospholipase Cγ (PLCγ) owing to their gene amplification or protein overexpression can lead to hyperactivation of the FGFR downstream signalling pathways.
GRB2, Growth factor receptor-bound protein 2.
*Samples from 4,853 cancers of various types were analysed for FGFR alterations on physician request. Urothelial carcinomas (UCs) include cancers of the renal pelvis (21 cases), ureter (6), bladder (90), and not otherwise specified (9). Gliomas include glioblastoma (84 cases), astrocytoma (21), ependymoma (7), oligodendroglioma (17), and glioma not otherwise specified (15).[1][6]